Principal investigator
Samuel Crish, Ph.D.
Associate Professor of Pharmaceutical Sciences
Contact
430 RGE
4209 State Route 44
Rootstown OH, 44272
330-325-6680

College of Pharmacy
Department of Pharmaceutical Sciences

Samuel Crish, Ph.D.
Associate Professor of Pharmaceutical Sciences
430 RGE
4209 State Route 44
Rootstown OH, 44272
330-325-6680
Our lab focuses on pre- and early degenerative defects in the visual system in glaucoma and Alzheimer’s disease (AD). Retinal ganglion cells (RGC), the projection cells of the eye that carry visual information to the brain, degenerate in both diseases. Once these cells are lost, visual signals cannot travel to the brain for further processing, resulting in blindness.
While most investigators either target death of the RGC body or try and regenerate new cells, we in the SCrish Lab take a different path. These diseases can take decades to blind. That is because neurons don’t just die overnight, it is a long process of “neuronal decline” — cells exhibit subtle changes to their structure and function that accumulate over years before irreversible RGC death. We believe that this extended decline presents a therapeutic window where restoring function to these “sick” neurons can slow or stop their journey to death. Maybe, in some cases, we can even reverse the process — rescuing damaged neurons that can get back to their jobs of carrying vision to the brain!
We have found several commonalities between the onset, progression, and mechanisms of neurodegeneration in glaucoma and Alzheimer’s disease. Our lab uses a combination of classical and cutting-edge methodologies such as electrophysiology, fluorescent and electron microscopy, advanced protein and gene expression quantitation techniques to tease out the mechanisms driving dysfunction and eventual neuronal death in these diseases. We then use that information to identify new targets of intervention and test new much-needed therapies for these devastating conditions.
such as impaired memory, other symptoms occur, including vision loss. Aggregations of the AD-associated proteins amyloid-beta (Aβ) and hyperphosphorylated tau (ptau) are evident in the retina up to a decade before cognitive decline. However, we do not know how these proteins impact vision. For this project, we inject different forms of these proteins directly into the eye and see how they drive retinal pathology and spread throughout the rest of the nervous system.
Below is an example of how injected Aβ kills retinal ganglion cells (RGC; white dots). Note the large areas of retina that have lost RGCs after Aβ injection. Below are fluorescent images of the retina showing Aβ being taken up by cells of the retina.

Neurodegeneration (neuron loss) and neuroinflammation (the nervous system’s immune response) are intimately entwined. Neuroinflammation drives neurodegeneration, which increases neuroinflammation, which drives more neurodegeneration, and it continues in this damaging cycle. Reducing neuroinflammation may be our best hope to stop the progression of pathology in neurodegenerations such as glaucoma. We are exploring the immunosuppresant multiple sclerosis drug fingolimod as a treatment for glaucoma. Our initial studies using fingolimod showed that one week of fingolimod treatment, restored damaged optic nerve fibers in a common mouse model of glaucoma, the DBA/2J mouse. We are currently testing whether this improvement improves other neuronal locations and restores defective eye-brain communication. Below is an image showing normal and defective optic nerve fibers and synapses before fingolimod treatment.

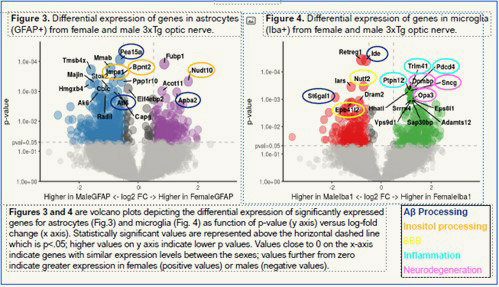
Alzheimer’s disease exhibits strong sex differences, with two-thirds of cases being women; they also have a much faster course of pathology. In collaboration with the CCrish Lab, we are exploring differences in visual system structure and function and using a bleeding-edge technique known as spatial transcriptomics to investigate differences in optic nerve glia (non-neuronal cells of the nervous system), which mediate both pathology and the response to damaging stressors. The below figure shows gene expression differences of over 12,000 genes in male and female optic nerves undergoing RGC damage with some common pathological pathways highlighted.
See open positions in this lab on the NEOMED careers site

Cheri Harris
Business Manager
Phone: 330.325.6689
Email: charris1@380cebbe0d.nxcli.io
Moses Oyewumi, Ph.D.
Phone: 330.325.6669
Email: moyewumi@380cebbe0d.nxcli.io

© 2024 Northeast Ohio Medical University
4209 St. Rt. 44, PO Box 95, Rootstown, Ohio 44272
ADA compliance | Consumer information | Title IX | Privacy statement | Do business with NEOMED