Principal investigator
Christine Dengler-Crish, Ph.D.
Assistant Professor, Department of Pharmaceutical Sciences

College of Pharmacy
Department of Pharmaceutical Sciences

Christine Dengler-Crish, Ph.D.
Assistant Professor, Department of Pharmaceutical Sciences
Our lab studies some of the earliest, yet overlooked factors that may be driving the neurodegenerative process of Alzheimer’s disease (AD) dementia. Our research philosophy is that alterations in the brain’s most fundamental abilities to sense and respond to information from the external world and bodily signals may be the key to understanding highly complex diseases like dementia. Increasing scientific evidence shows that the brain’s ability to process sensory information and regulate bodily homeostasis are compromised in AD prior to the first signs of memory loss. However, these neural processes are often overlooked because dementia is classically considered to be a disorder of cognition and memory. This is problematic to the field because there is a critical need to identify and deploy early interventions in AD to halt this devastating disease before cognition, memory, and personality are irreparably destroyed. Our research investigates two major areas: a) mechanisms by which visual system function is disrupted in AD and may further contribute to brain damage and cognitive decline, and b) how disruptions in bodily homeostasis such as bone loss or altered energy metabolism may indicate that the dementia disease process is beginning. In studying how these early changes in body and brain function contribute to dementia, we hope to identify new strategies and treatments that can be used to prevent the onset and progression of AD and spare patients and their families the devastating consequences of this fatal disease.
We are studying factors that contribute to neurofibrillary tau pathogenesis (i.e. tauopathy), a hallmark pathology of dementia that accumulates in preclinical disease stages and correlates closely with clinical progression of cognitive decline in dementia. We recently found that that the exercise-induced hormone irisin—which is known to modulate critical cell surface proteins called integrins– reduces tau pathology and inflammation in the brains of transgenic tauopathy-model htau mice in a sex and age-dependent manner. We are currently testing whether integrin function is altered by AD risk factors or susceptibility genes, and whether this causes integrins to activate inflammatory and neurodegenerative pathways that promote onset and progression of tauopathy.
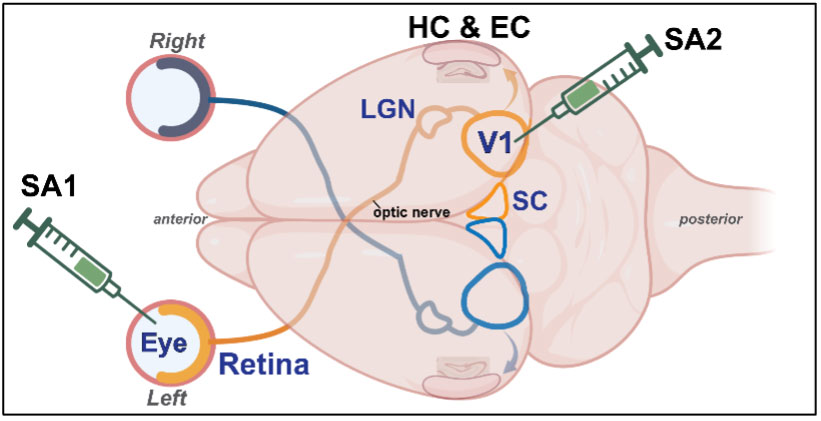
We are investigating how presence of pathological Aβ or tau proteins in the retina causes damaging changes along the primary retinal pathway to primary visual cortex (V1), and whether disease mechanisms can then spread via V1’s interconnections to brain regions affecting learning and memory. To study this, we are experimentally injecting Aβ or tau and proteins into either the eyes or V1 of healthy mice and measuring the effects of this manipulation on visual system circuitry using a combination of electrophysiological and immunohistochemical techniques.
See open positions in this lab on the NEOMED careers site.
College of Pharmacy
College of Medicine
Our research has been funded by NIA/NIH, the Alzheimer’s Association, the Ohio Lions Eye Research Foundation, Prevent Blindness Ohio, the Templeton Medical Research Foundation, and the Alan and Janice Woll Family Foundation.
Cheri Harris
Business Manager
Phone: 330.325.6689
Email: charris1@380cebbe0d.nxcli.io
Moses Oyewumi, Ph.D.
Phone: 330.325.6669
Email: moyewumi@380cebbe0d.nxcli.io

© 2024 Northeast Ohio Medical University
4209 St. Rt. 44, PO Box 95, Rootstown, Ohio 44272
ADA compliance | Consumer information | Title IX | Privacy statement | Do business with NEOMED